Standard Enthalpy Of Formation Of Ammonia. See all problems in enthalpy of formation. What is the standard enthalpy of formation of ammonia? The standard enthalpy of formation of nh3 is 460 kjmol if the enthalpy of formation of h2 from its atoms is 436 kjmol and that of n2 is 712 kjmol the average bond enthalpy of n h bond in nh3 is a 1102. A = 6.67956, b = 1002.711, c = 25.215 for t = 190 k through 333 k.
 Nitrogen Gas Nitrogen Gas Enthalpy Of Formation From nitrogengassanroso.blogspot.com
Nitrogen Gas Nitrogen Gas Enthalpy Of Formation From nitrogengassanroso.blogspot.com
16 rows selected atct [1, 2] enthalpy of formation based on version 1.118 of the. All elements in their standard states have a standard. However, the enthalpy of formation of ammonia definition requires that only 1 mole of ammonia is formed. ½n 2 (g) + h 2 (g) nh 3 (g) clearly, the energy change of formation of ammonia is half that of the enthalpy change of reaction for the balanced equation. For which of the following standard heat of formation is. All chemistry practice problems enthalpy of formation practice problems.
What is the standard enthalpy of formation of ammonia?
The standard enthalpy of formation (δ f h 2 9 8 o ) for methane, c h 4 is − 7 4. What is the standard enthalpy of formation of ammonia? Subscribe and get access to thousands of top quality interact. C 2 h 6 (g) + 7f 2 (g) 2cf 4 (g) + 6hf(g) use this value and the standard enthalpies of formation in table 1 to calculate the The standard enthalpy of formation of ammonia is calculated as: Click here👆to get an answer to your question ️ the standard enthalpy of formation of ammonia gas isgiven :
 Source: youtube.com
Source: youtube.com
Therefore the standard dh of formation. 9 k j m o l − 1. (c) table 1 gives some values of standard enthalpies of formation (δ f hθ). Therefore the standard dh of formation. Standard enthalpy of formation* for atomic and molecular ions cations δh˚ f (kj/mol) cations δh˚ f (kj/mol) anions δh˚ f (kj/mol) anions δh˚ f (kj/mol) ag+(aq) +105.9 k+(aq) −251.2 br−(aq) −120.9 h 2po 4 −(aq) −1302.5 al3+(aq) −524.7 li+(aq) −278.5 cl−(aq) −167.4 hpo 4 2−(aq) −1298.7 ba2+(aq) −538.4 mg2+(aq) −462.0 clo
 Source: chegg.com
Source: chegg.com
The energy spent will be +92.0 kj mol −1 since the described reaction implies the breakdown of two moles of ammonia. The chemical reaction for the formation of. 4nh3 (g) + 7o2 (g) → 4no2 (g) + 6h2o (l) using standard molar enthalpies of formation. A = 6.67956, b = 1002.711, c = 25.215 for t = 190 k through 333 k. The standard enthalpy of formation (δ f h 2 9 8 o ) for methane, c h 4 is − 7 4.
 Source: homeworklib.com
Source: homeworklib.com
Δh (reaction) = 2 x δhf (nh 3) enthalpy change. Subscribe and get access to thousands of top quality interact. Learn this topic by watching enthalpy of formation concept videos. 16 rows selected atct [1, 2] enthalpy of formation based on version 1.118 of the. The standard enthalpy of formation is measured in units of energy per amount of substance, usually stated in kilojoule per mole, but also in kilocalorie per mole, joule per mole or kilocalorie per gram.
 Source: homeworklib.com
Source: homeworklib.com
Table 1 substance f 2 (g) cf 4 (g) hf(g) δ f hθ / kj mol−1 0 −680 −269 the enthalpy change for the following reaction is −2889 kj mol−1. A = 6.67956, b = 1002.711, c = 25.215 for t = 190 k through 333 k. Calculate the ∆rh° for the combustion of ammonia: Subscribe and get access to thousands of top quality interact. Calculate δh o f for ammonia.
 Source: tessshebaylo.com
Source: tessshebaylo.com
C 2 h 6 (g) + 7f 2 (g) 2cf 4 (g) + 6hf(g) use this value and the standard enthalpies of formation in table 1 to calculate the C 2 h 6 (g) + 7f 2 (g) 2cf 4 (g) + 6hf(g) use this value and the standard enthalpies of formation in table 1 to calculate the 4nh3 (g) + 7o2 (g) → 4no2 (g) + 6h2o (l) using standard molar enthalpies of formation. The standard enthalpy of formation (δ f h 2 9 8 o ) for methane, c h 4 is − 7 4. Thus in the reaction the enthalpy of formation 2 mole of nh3 is 92kj.
 Source: chegg.com
Source: chegg.com
Because the formation of 1 mole of ammonia releases −46.0 kj mol −1 of energy, the breakdown of 1 mole of ammonia consumes the same amount of energy, i.e. The standard enthalpy of formation of ammonia is calculated as: A = 6.67956, b = 1002.711, c = 25.215 for t = 190 k through 333 k. The standard enthalpies of formation are: Click here👆to get an answer to your question ️ the standard enthalpy of formation of ammonia gas isgiven :
 Source: slidesharetips.blogspot.com
Source: slidesharetips.blogspot.com
In order to calculate the average energy given out in the formation of a c − h bond from this, it is necessary to know which one of the following? C 2 h 6 (g) + 7f 2 (g) 2cf 4 (g) + 6hf(g) use this value and the standard enthalpies of formation in table 1 to calculate the See all problems in enthalpy of formation. 16 rows selected atct [1, 2] enthalpy of formation based on version 1.118 of the. The standard enthalpy of formation is measured in units of energy per amount of substance, usually stated in kilojoule per mole, but also in kilocalorie per mole, joule per mole or kilocalorie per gram.
 Source: tardigrade.in
Source: tardigrade.in
The standard enthalpy of formation of ammonia is calculated as: Learn this topic by watching enthalpy of formation concept videos. C 2 h 6 (g) + 7f 2 (g) 2cf 4 (g) + 6hf(g) use this value and the standard enthalpies of formation in table 1 to calculate the A = 6.67956, b = 1002.711, c = 25.215 for t = 190 k through 333 k. The standard enthalpy of formation of ammonia is calculated as:
 Source: youtube.com
Source: youtube.com
For which of the following standard heat of formation is. Subscribe and get access to thousands of top quality interact. ½n 2 (g) + h 2 (g) nh 3 (g) clearly, the energy change of formation of ammonia is half that of the enthalpy change of reaction for the balanced equation. All chemistry practice problems enthalpy of formation practice problems. The standard enthalpy of formation of ammonia is calculated as:
 Source: nitrogengassanroso.blogspot.com
Source: nitrogengassanroso.blogspot.com
Click here👆to get an answer to your question ️ the standard enthalpy of formation of ammonia gas isgiven : ½n 2 (g) + h 2 (g) nh 3 (g) clearly, the energy change of formation of ammonia is half that of the enthalpy change of reaction for the balanced equation. The standard enthalpy of formation (δ f h 2 9 8 o ) for methane, c h 4 is − 7 4. The results were compared with values from other sources, and the value so (nh 4+, aq, 298.15 k) = (26.45 ± 0.13) cal th k −1 mol −1 was calculated from. Therefore the standard dh of formation.
 Source: chegg.com
Source: chegg.com
The standard enthalpy of solution of gaseous ammonia in water to form the non. The standard enthalpies of formation are: Because the formation of 1 mole of ammonia releases −46.0 kj mol −1 of energy, the breakdown of 1 mole of ammonia consumes the same amount of energy, i.e. See all problems in enthalpy of formation. C 2 h 6 (g) + 7f 2 (g) 2cf 4 (g) + 6hf(g) use this value and the standard enthalpies of formation in table 1 to calculate the
 Source: slideshare.net
Source: slideshare.net
What is the standard enthalpy of formation of ammonia? Thus in the reaction the enthalpy of formation 2 mole of nh3 is 92kj. Click here👆to get an answer to your question ️ the standard enthalpy of formation of ammonia gas isgiven : From the table of values for standard enthalpy of formation at 25°c given in the previous section, we find that the standard enthalpy of formation of ammonium chloride (nh 4 cl (s)) is. The enthalpy change for the reaction 2nh3 (g) → n2 (g) + 3h2 (g) is:

Source: youtube.com
The standard enthalpy of solution of gaseous ammonia in water to form the non. All elements in their standard states have a standard. For which of the following standard heat of formation is. In order to calculate the average energy given out in the formation of a c − h bond from this, it is necessary to know which one of the following? (c) table 1 gives some values of standard enthalpies of formation (δ f hθ).
 Source: homeworklib.com
Source: homeworklib.com
½n 2 (g) + h 2 (g) nh 3 (g) clearly, the energy change of formation of ammonia is half that of the enthalpy change of reaction for the balanced equation. For which of the following standard heat of formation is. Table 1 substance f 2 (g) cf 4 (g) hf(g) δ f hθ / kj mol−1 0 −680 −269 the enthalpy change for the following reaction is −2889 kj mol−1. The standard enthalpy of solution of gaseous ammonia in water to form the non. Therefore the standard dh of formation.
 Source: homeworklib.com
Source: homeworklib.com
All elements in their standard states have a standard. The standard enthalpy of formation of ammonia is calculated as: From the table of values for standard enthalpy of formation at 25°c given in the previous section, we find that the standard enthalpy of formation of ammonium chloride (nh 4 cl (s)) is. The enthalpy change for the reaction 2nh3 (g) → n2 (g) + 3h2 (g) is: The standard enthalpy of solution of gaseous ammonia in water to form the non.
 Source: slideshare.net
Source: slideshare.net
In order to calculate the average energy given out in the formation of a c − h bond from this, it is necessary to know which one of the following? The chemical reaction for the formation of. Standard enthalpy of formation* for atomic and molecular ions cations δh˚ f (kj/mol) cations δh˚ f (kj/mol) anions δh˚ f (kj/mol) anions δh˚ f (kj/mol) ag+(aq) +105.9 k+(aq) −251.2 br−(aq) −120.9 h 2po 4 −(aq) −1302.5 al3+(aq) −524.7 li+(aq) −278.5 cl−(aq) −167.4 hpo 4 2−(aq) −1298.7 ba2+(aq) −538.4 mg2+(aq) −462.0 clo A = 6.67956, b = 1002.711, c = 25.215 for t = 190 k through 333 k. Click here👆to get an answer to your question ️ the standard enthalpy of formation of ammonia gas isgiven :

Standard enthalpy of formation* for atomic and molecular ions cations δh˚ f (kj/mol) cations δh˚ f (kj/mol) anions δh˚ f (kj/mol) anions δh˚ f (kj/mol) ag+(aq) +105.9 k+(aq) −251.2 br−(aq) −120.9 h 2po 4 −(aq) −1302.5 al3+(aq) −524.7 li+(aq) −278.5 cl−(aq) −167.4 hpo 4 2−(aq) −1298.7 ba2+(aq) −538.4 mg2+(aq) −462.0 clo 16 rows selected atct [1, 2] enthalpy of formation based on version 1.118 of the. The standard enthalpy of formation of nh3 is 460 kjmol if the enthalpy of formation of h2 from its atoms is 436 kjmol and that of n2 is 712 kjmol the average bond enthalpy of n h bond in nh3 is a 1102. ½n 2 (g) + h 2 (g) nh 3 (g) clearly, the energy change of formation of ammonia is half that of the enthalpy change of reaction for the balanced equation. Subscribe and get access to thousands of top quality interact.
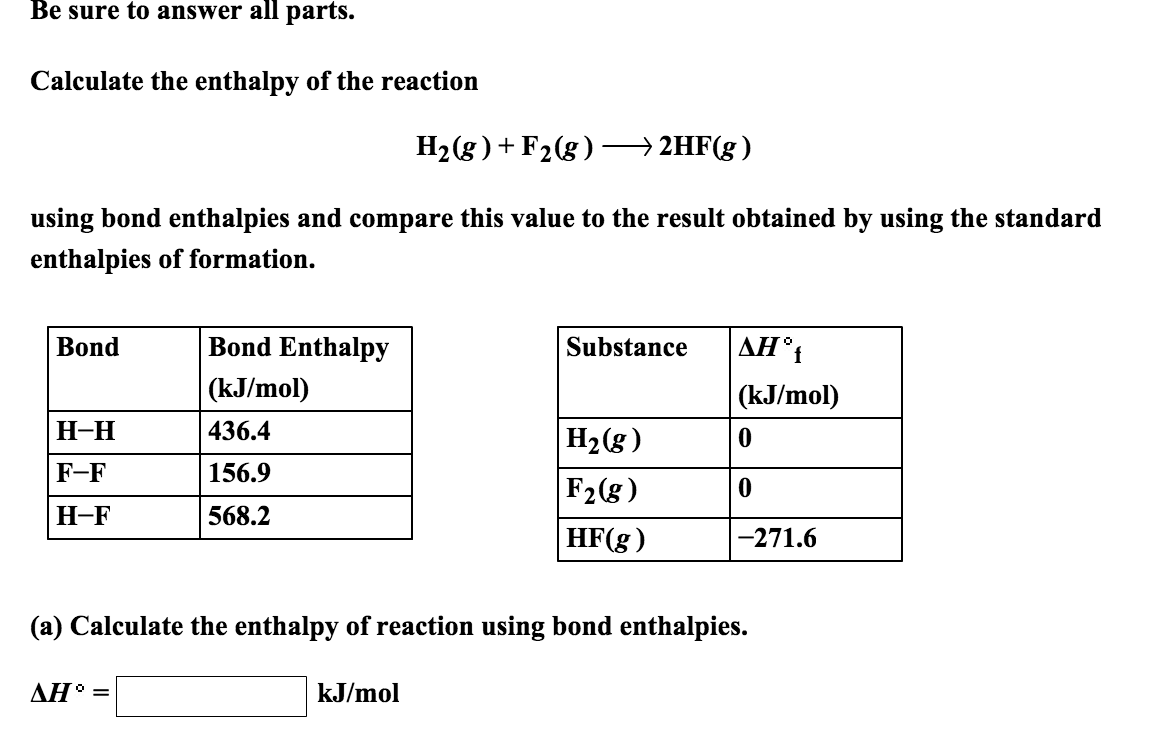 Source: chegg.com
Source: chegg.com
In order to calculate the average energy given out in the formation of a c − h bond from this, it is necessary to know which one of the following? 9 k j m o l − 1. Thus in the reaction the enthalpy of formation 2 mole of nh3 is 92kj. However, the enthalpy of formation of ammonia definition requires that only 1 mole of ammonia is formed. ½n 2 (g) + h 2 (g) nh 3 (g) clearly, the energy change of formation of ammonia is half that of the enthalpy change of reaction for the balanced equation.
This site is an open community for users to do submittion their favorite wallpapers on the internet, all images or pictures in this website are for personal wallpaper use only, it is stricly prohibited to use this wallpaper for commercial purposes, if you are the author and find this image is shared without your permission, please kindly raise a DMCA report to Us.
If you find this site convienient, please support us by sharing this posts to your own social media accounts like Facebook, Instagram and so on or you can also bookmark this blog page with the title standard enthalpy of formation of ammonia by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.




